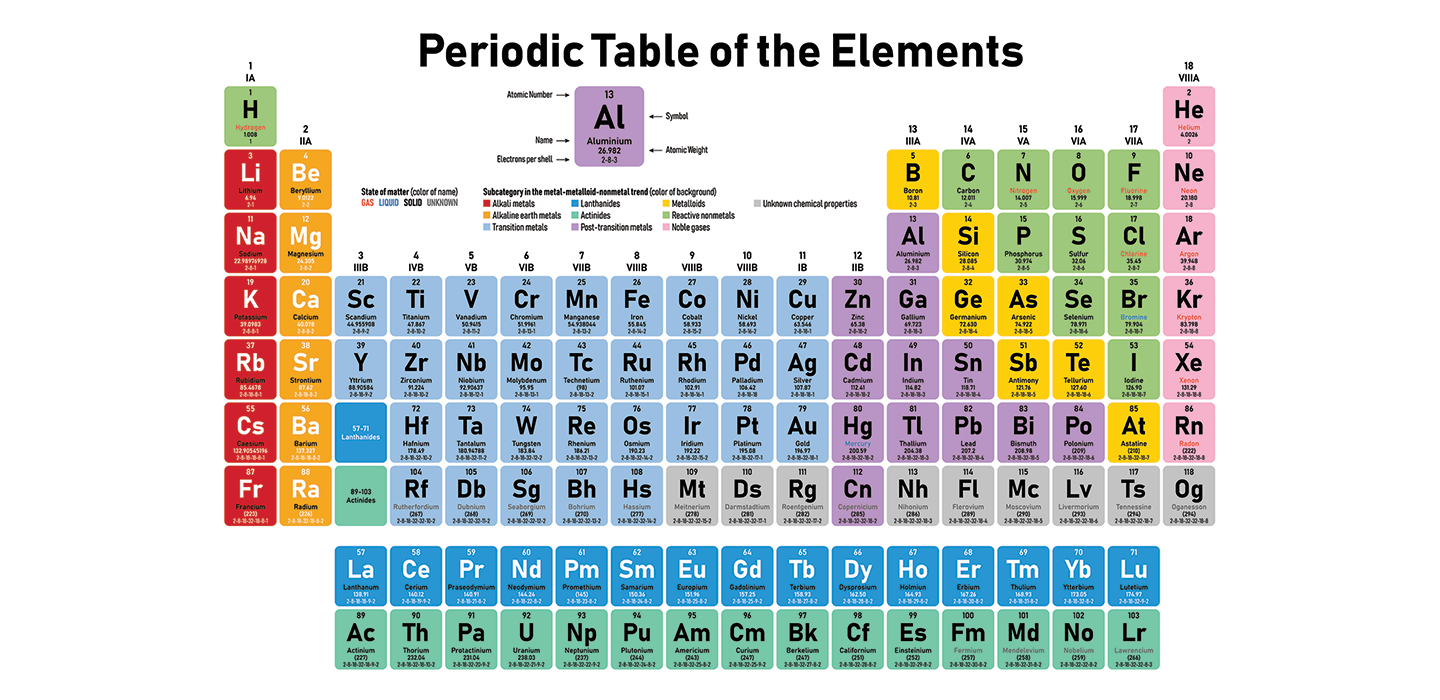
It is the most electronegative element, given that it is the top element in the Halogen Group, and therefore is very reactive. The periodic table lists the atomic weight for each element, which can be used to find mass number, For hydrogen, for example, the atomic weight is 1.008. Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons. Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons. Its atomic number is 9 and its atomic weight is 19, and it's a gas at room temperature. Another common method of categorization recognizes nine element families: Alkali Metals: Group 1 (IA) - 1 valence electron. German chemists Ida Tacke and Walter Noddack set out to find the elements, a quest being pursued by scientists around the world. Chemistry of Fluorine (Z9) Fluorine (F) is the first element in the Halogen group (group 17) in the periodic table. Mendeleev had predicted, and others including Henry Moseley had later confirmed, that there should be elements below Manganese in Group 7. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 1–18 from left to right) the seven horizontal rows are called periods.\): Elements in the periodic table are organized according to their properties.Įven after the periodic nature of elements and the table itself were widely accepted, gaps remained. It arranges of the elements in order of increasing atomic number. Even though it is in the same column or group of the periodic table with the other halogens, most. Therefore, the resultant ion is symbolized as I. Because this element is located in Group 17, or 7A, on the periodic table, it will ionize to form an anion with a 1 charge. Element 117, tennessine, might have some properties in common with the other elements. Recall that all elements found within the same column on the periodic table have the same number of valence electrons. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Fluorine, chlorine, bromine, iodine, and astatine definitely are halogens. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.The chemical symbol for Hydrogen is H. 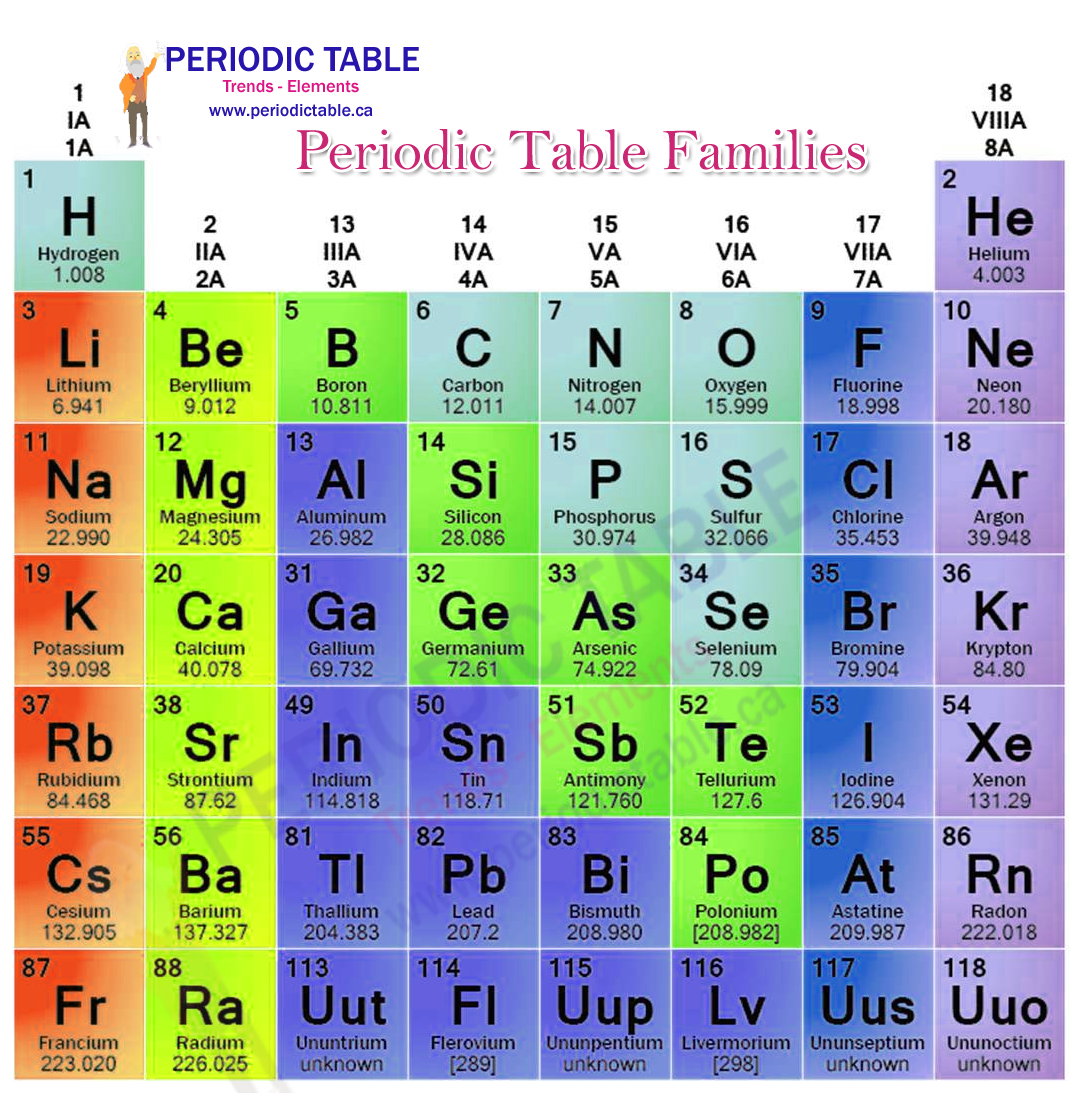
The periodic table is used as a predictive tool. Depending on who you ask, there are either 5 or 6 halogens. As expected, semimetals exhibit properties intermediate between metals and nonmetals. The Periodic Table has 118 elements which organized on the basis of atomic number and grouped based on similarity in chemical properties. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Chlorine is a chemical element of the periodic table with chemical symbol Cl and atomic number 17 with an atomic weight of 35. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The elements are arranged in seven horizontal. (a) What is the most common oxidation state. Each box represents an element and contains its atomic number, symbol, average atomic mass, and (sometimes) name. Elements in group 17 in the periodic table are called the halogens elements in group 16 are called the chalcogens. The vast majority of the known elements are metals. A modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column (Figure 3.2.2 3.2. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. A list of reference sources used to compile the data provided on our periodic. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Atomic Number: 17 Group: 17 Period: 3 Series: Halogens. It is also used in the production of many plastics such as PVC. It is used in swimming pools commonly and also making drinking water safe by killing bacteria. Chlorine much like other Halogens has antiseptic properties. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. It has the atomic number 17 in the periodic table and belongs in Group 17, the Halogens. 
\) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
